《鄉民大學問EP.38》字幕版|藍營2028的超級戰術!黃暐瀚:民調洩民進黨這弱點!廢死引爆民怨 是蔡英文挖坑給賴清德?台電屢跳電 萬美玲曝內幕:竟是小鳥惹的禍?!賴清德赴立院國情報告?將對上韓國瑜?
NOW影音
更多NOW影音焦點
更多焦點-

炎黃子孫是古代傳說?國台辦批民進黨褻瀆 陸委會:百科網站講的
前總統馬英九4月初訪問中國後返台,喊炎黃子孫同屬中華民國。陸委會對此回應,炎黃子孫只是神話,引發國台辦不滿,稱這是「鐵一般的事實不容否認、不容褻瀆」。陸委會昨再回應,兩岸的百科網站都是這樣講。國台辦發
2024-04-26 15:46
-

聯發科Q1 EPS 19.85元、單季歷史第三高!毛利率52.4%超標
聯發科今(26)日召開法說會,首季獲利為316.55億元,季增23.1%、年增87.4%,單季每股純益為19.85元,以歷年同期的表現來看,為單季歷史第三高。聯發科首季合併營收為1334.58億元,季
2024-04-26 15:32
-

獨/神老師氣到發抖嗆林叨囝仔7寶媽!分享育兒經驗:不要看現在
「林叨囝仔The Lins'Kids」7寶媽Sydney,因直播中出現疑似訕笑「資源班」學生言論而遭炎上,對此,網紅作家「神老師&神媽咪」沈雅琪作為老師及特殊孩子的媽媽,忍無可忍發聲痛批:「憑
2024-04-26 12:36
-
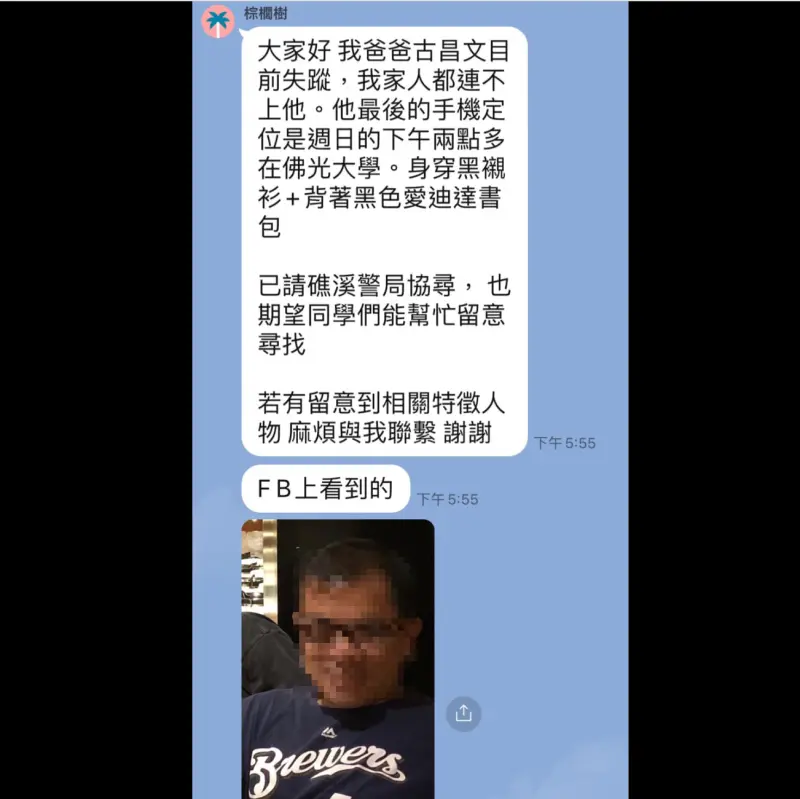
花蓮醫定位在山區人間蒸發!賞鯨船上留「名片+背包」 家屬急尋
離奇失蹤案件!花蓮慈濟醫院一名古姓醫師,21日前往礁溪參加醫學研討會,豈料家屬卻遲遲聯絡不到人,基地台最後定位顯示,古姓醫師曾出現在當地山區,當時古男曾到烏石港搭船賞鯨,不過搭乘的船隻到港時,他已不見
2024-04-26 15:03
精選專題
要聞
更多要聞-

炎黃子孫是古代傳說?國台辦批民進黨褻瀆 陸委會:百科網站講的
前總統馬英九4月初訪問中國後返台,喊炎黃子孫同屬中華民國。陸委會對此回應,炎黃子孫只是神話,引發國台辦不滿,稱這是「鐵一般的事實不容否認、不容褻瀆」。陸委會昨再回應,兩岸的百科網站都是這樣講。國台辦發
2024-04-26 15:46
-

李正皓贊助特教生奪世界冠軍 「孩子應平等對待」嗆爆「七寶媽」
網紅「七寶媽」近日直播時帶兒子共同訕笑資源班特教生,引發眾怒,多數廠商也紛紛與她切割。對此,時事評論員李正皓今(26)日在臉書PO出一段影片,是來自苗栗特殊教育學校啦啦隊赴美參賽,勇奪世界冠軍,直言「
2024-04-26 15:32
-

國民黨內鬥又起?傳侯友宜選黨魁 他批:2026埋下「掉2都」危機
國民黨主席朱立倫明年10月卸任,明天將進入國民黨主席選戰,有媒體報導,新北市長侯友宜可能參選黨主席。但事實上,侯友宜並未擔任過中央委員,目前並無參選黨主席資格。資深媒體人韋安說,國民黨亂鬥又起不知死活
2024-04-26 15:28
-

曝展開「第三代計畫」 柯文哲談2028:2026選很差就可以收攤了
民眾黨主席柯文哲2024挑戰總統大位失利,曾表示4年後要再拚一次,柯文哲今(26)日上節目《觀點芹爆戰》,透露自己已開始「第三代計畫」,而2026決定2028,「2026選很差就可以收攤了。」柯文哲指
2024-04-26 15:24
新奇
更多新奇-

台灣泡麵「隱藏王牌」公開!每碗37元內行扛整箱 眾吃20年沒看過
台灣泡麵在市面上販售的口味非常眾多,除了定期的新品之外,也有許多老字號的泡麵長年稱霸排行榜,成為台灣泡麵的一大特色。然而,近日就有網友分享一款台灣泡麵隱藏版,由於平常沒有在各大通路上架,因此只有內行人
2024-04-26 14:20
-

七寶媽學歷曝!從小是學霸「最強外語」大學畢 自嘲專長:生小孩
35歲的網紅七寶媽(陳珮芬)歧視資源班言論遭炎上,不少人好奇七寶媽學歷為何?「七寶媽 學歷」在Google熱搜中快速飆升。據悉,七寶媽從小就是個學霸,國小到大學成績都名列前茅,她曾在部落格自曝畢業於文
2024-04-26 11:26
-

婦產科蔡醫師迷因爆紅!本尊是世界名醫 粉絲讚爆:我是試管嬰兒
2024最新迷因爆紅!最近在社群平台上,出現一名「活體迷因」,一位婦產科蔡醫師大爆紅,不僅有IG、Tik Tok的追蹤人數暴衝,甚至還有許多粉絲做了蔡醫師的各種周邊商品,由於過去蔡醫師還開投票讓粉絲表
2024-04-26 11:13
-

快訊/網紅七寶媽笑資源班遭轟!刪直播二度道歉:請原諒我的無知
網紅「林叨囝仔The Lins'Kids」七寶媽去年陷入多起爭議,好不容易才悄悄復出,結果日前直播上疑似嘲笑資源班的言論,讓她再度炎上。日前七寶媽Sydney深夜開直播道歉,但卻被批評是「把過
2024-04-26 09:15
娛樂
更多娛樂-

《雲之羽》女星證實未婚懷孕!金靖曝工作想吐「靠大笑掩飾害喜」
去年因熱播陸劇《雲之羽》受矚目的31歲大陸女星金靖,昨(25)日突發文自爆已懷孕,「一家四口一起迎接第五位家庭成员」,她也透露另一半為導演舒奕橙。金靖8年前以綜藝節目《今夜百樂門》出道,個性爽朗的她自
2024-04-26 15:46
-

鄭靚歆女女大婚!美魔女媽「透明薄紗內衣全露」 母女前晚爆爭執
28歲鄭靚歆(Jin)從節目《我愛黑澀會》出道,參加《全明星運動會4》收獲不少粉絲,她去年6月和大5歲德國女友采熙(Aky)登記結婚,今(26)日終於舉辦婚宴,鄭靚歆的美魔女媽媽胡文英透視薄紗裡只穿比
2024-04-26 15:29
-

啦啦隊一粒獲「輔大校花認證」!學生時期照曝光 貓耳造型超可愛
台鋼啦啦隊成員一粒(本名趙宜莉)因為其深邃的雙眼及高挺的鼻樑,再加上她的「葉保弟應援曲」魔性舞步。洗腦的歌曲加上好記的舞步,讓不少啦啦隊、網紅、藝人爭相模仿一粒的魔性舞步,就連日本AV女優也跟著模仿。
2024-04-26 15:11
-

林叨囝仔7寶媽再惹議!視網膜經紀韓璟曝國小唸過:高中讀資優班
在臉書上擁有30萬人追蹤的網紅「林叨囝仔The Lins'Kids」7寶媽,近期在直播時因嘲笑、歧視資源班學生,引來社會大眾批評,目前已經有10家廠商宣布與她做切割。對此,前立委助理、現任網紅
2024-04-26 14:39
運動
更多運動-

Wembanyama防守有多恐怖?Wilkins盛讚:就算沒蓋鍋也影響了出手
聖安東尼奧馬刺超級新星Victor Wembanyama在生涯首季就繳出劃時代的數據,他在阻攻上的天賦,被看好競爭本季的年度最佳防守球員(DPOY)。近期來到台灣參加「2024 國泰NBA國際高中邀請
2024-04-26 15:45
-

NBA季後賽4/26戰報/湖人主場吞絕望一敗 金塊首輪3:0搶先聽牌
NBA在4月26日進行3場首輪季後賽的賽事,其中最受矚目的一場,無疑就是丹佛金塊在客場以112:105擊敗洛杉磯湖人,系列賽以3:0領先,成為本季首輪第一支「聽牌」的球隊。該場比賽「最強亞軍」Aaro
2024-04-26 15:33
-

湖人「變招」失敗 AD出禁區、讓Aaron Gordon活生生打成了歐尼爾
這是洛杉磯湖人在這個系列賽輸得最簡單的一場比賽嗎?今(26)日他們以105:112不敵丹佛金塊,球隊在第三節就繳械投降了,金塊只是利用Aaron Gordon的體型就輕鬆逆轉了戰局,此役的失利說明了湖
2024-04-26 15:15
-

徐基麟挑戰陳義信大紀錄還要再等等!中信兄弟、統一獅宣布延賽
中信兄弟今(26)日進行統一獅與中信兄弟之戰,不過因為台中降雨影響,比賽也被迫延期,中職今天三地開打,沒想到都因雨延賽。原訂中信定先發投手徐基麟,也因延賽無法搶第四勝,過去在兄弟隊史上,曾有4位投手達
2024-04-26 15:12
財經生活
更多財經生活-

快訊/暴雨橫掃南部!高雄市1國小下午停班課 豪雨假標準一次看
鋒面挾帶強對流!中南部雷雨不斷,從25日0時至今(26)日下午13時10分,高雄鼓山累積雨量達193.5毫米,高雄市政府宣布,由於山區大雨,考量師生安全,桃源區寶山國民小學自15時起停止上班、停止上課
2024-04-26 15:22
-
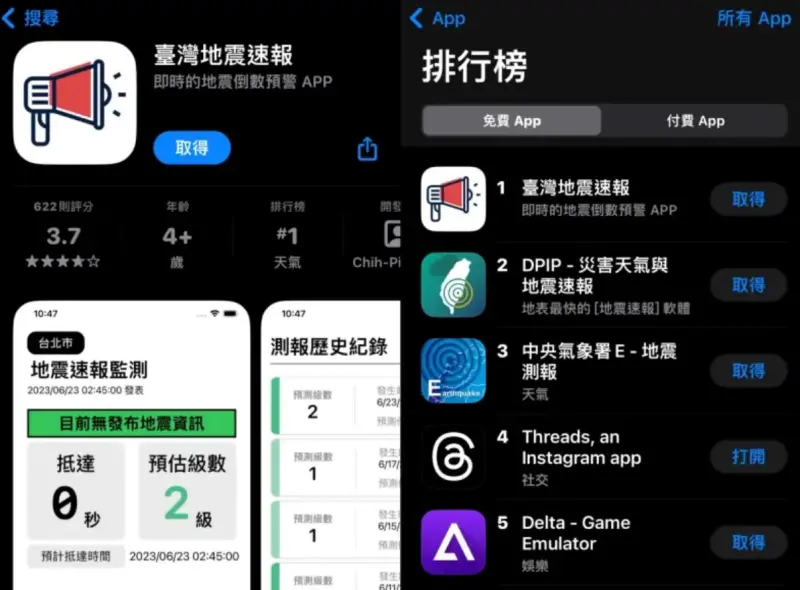
「地震速報App」高中生深夜致歉!氣象署澄清:未以授權角度關切
0403花蓮強震後餘震不斷,民眾對於地震相關APP關注度提高,其中一款林子祐同學開發的「台灣地震速報」App受到民眾喜愛,獲得大量下載以及關注,不過由於數據是靠「地牛Wake Up」間接取得中央氣象署
2024-04-26 14:43
-

7寶媽嘲笑資源班慘了!第10間廠商發聲切割 康軒:遠離歧視言論
網紅「林叨囝仔The Lins'Kids」7寶媽Sydney日前在直播時,被質疑恥笑、歧視「資源班」學生,失言言論立刻遭到炎上,雖然7寶媽Sydney在昨(25)日公開道歉,但仍難以平息眾人怒
2024-04-26 14:23
-

雷雨胞持續移入!南部降雨達194毫米 彭啟明:解渴致災一線之隔
鋒面挾帶強對流持續移入台灣上空,中南部雷雨不斷,從25日0時至今(26)日下午13時10分,高雄鼓山累積雨量達193.5毫米,台南、嘉義部分地區也在140毫米上下。氣象專家彭啟明表示,雷雨胞持續移入,
2024-04-26 13:46
全球
更多全球-

挺巴活動蔓延全美!民調:搖擺州選民 越來越不支持援助以色列
美國多間大學聲援巴勒斯坦的示威潮,近日越演越烈,據悉已有數百名學生被捕,陸續都有學校因考量安全,宣布原訂5月舉行的畢業典禮。而據外媒1份最新民調顯示,自從以色列開始密集空襲加薩以來,搖擺州選民對美國援
2024-04-26 15:13
-

櫻花釀禍?日本知名賞櫻景點現大量魚屍 居民怕:擔心有毒物流入
日本賞櫻季節邁入尾聲,不過隨著花瓣飄落、綠葉冒出,卻出現了驚人景象。近來有日本民眾發現,東京知名賞櫻景點目黑川當中,可以看到魚屍跟花瓣一同在水面上飄蕩,且目擊到魚屍的地點還不只一處,讓附近居民十分擔心
2024-04-26 15:01
-

站著說話不腰疼?中國專家籲年輕人「別啃老」 網諷:養老金太多
中國經濟陷入困局,受到高房價、高失業率影響,「買房」對中國年輕人來說已經是遙不可及的夢想。但近日,中國一名經濟學專家公開呼籲年輕人,在買房的問題上不要焦慮,應該老實租房、不要啃老。這番言論引起網路討論
2024-04-26 12:37
-

波音出包、財務吃緊!美西南航空結束4機場服務 2千人丟飯碗
美國西南航空(Southwest Airlines)表示,由於波音(Boeing)延誤交付新飛機,導致公司成本上升,加上第一季財務虧損增加,將於今年底裁員2000人、結束4個機場的服務。根據《CNN》
2024-04-26 12:13